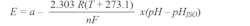A pH Sensor Reference Design Enabled for Wireless Transmission (Part 1)
Download this article in PDF format.
The first part of this article series addresses the pH probe and an examination of the different design challenges related to the front-end signal-conditioning circuit, including how to achieve low cost with high precision and reliability in data conversion. Part 2 delves into increasing the accuracy and precision in data processing, discussing calibration techniques such as a general polynomial fit using the least-square method for approximation in scattered predefined data for pH calibration. It will also provide a reference circuit design for a wireless monitoring system.
Understanding the pH Probe
An aqueous solution can fall under acidic, alkaline, or neutral levels. In chemistry, this is measured by a numeric scale, called pH, which stands for power of hydrogen according to the Carlsberg Foundation. This scale is logarithmic and goes from 1 to 14. The pH level can be expressed mathematically as pH = −log (H+). Therefore, if the hydrogen ion concentration is 1.0 × 10−2 moles/liter, then pH = −log (1.0 × 10−2) gives a value of 2.
An aqueous solution like distilled water has a pH value of 7, which is a neutral value. Solutions with a pH value below 7 are acidic, while those above a pH value of 7 are considered an alkaline solution. The logarithmic scale gives a degree of the acidity of a solution as compared to another.
For example, a solution with a pH value of 5 is 10 times more acidic than a solution with a pH value of 6, and 1,000 times more acidic than a solution with a pH value of 8. There are many ways to measure the pH level of an aqueous solution—it can be done through a litmus-paper indicator or via a glass probe.
Litmus Paper
A litmus-paper indicator is usually made up of dyes extracted from lichens that serve as a pH-level indicator. Once in contact with a solution, the paper has a chemical reaction that results in a change in color indicating the pH level.
This category basically includes two methods: one involves comparing the standard color corresponding to a known pH with the color of an indicator immersed in the test liquid using buffer solution. The other method involves preparing pH test paper that’s soaked in the indicator, then immersing the paper in the test liquid and comparing its color with the standard color. Though these two methods are easy to implement, they can be prone to errors resulting from temperature and foreign substances in the test solution.
pH Glass Probe
The most commonly used pH indicator is a pH probe. It consists of a glass measuring electrode and a reference electrode. A typical glass probe is made up of a thin glass membrane that encloses a hydrogen-chloride (HCl) solution. Inside the enclosure is a silver wire coated with AgCl, which acts as a reference electrode that’s in contact with the HCl solution. The hydrogen ions outside the glass membrane diffuse through the glass membrane and displace a corresponding number of sodium ions (Na+), which are normally present in most glasses. This positive ion is subtle and confined mostly to the glass surface on whichever side of the membrane has the lower concentration. The excess charge from the Na+ generates a potential voltage at the sensor output.
The probe is analogous to a battery. When the probe is placed in a solution, the measuring electrode generates a voltage depending on the hydrogen activity in the solution, which is compared to the potential of the reference electrode. As the solution becomes more acidic (lower pH value), the potential of the glass electrode becomes more positive (+mV) in comparison to the reference electrode.
As the solution becomes more alkaline (higher pH value), the potential of the glass electrode becomes more negative (−mV) in comparison to the reference electrode. The difference between these two electrodes is the measured potential. A typical pH probe ideally produces 59.154 mV/pH units at 25°C. This is usually expressed by the Nernst equation as shown below:
where:
E = voltage of the hydrogen electrode with unknown activity a = ±30 mV, zero-point tolerance
T = ambient temperature in 25°C
n = 1 at 25°C, valence (number of charges on ion) F = 96485 coulombs/mol, Faraday constant
R = 8.314 volt-coulombs/°K mol, Avogadro’s number
pH = hydrogen ion concentration of an unknown solution pHISO = 7, reference hydrogen ion concentration
The equation shows that the voltage generated depends on the acidity or alkalinity of the solution and varies with the hydrogen ion activity in a known manner. The change in temperature of the solution changes the activity of its hydrogen ions. When the solution is heated, the hydrogen ions move faster, which results in an increase in potential difference across the two electrodes. When the solution is cooled, the hydrogen activity decreases, causing a decrease in the potential difference. Electrodes are designed ideally to produce a zero-volt potential when placed in a buffer solution with a pH value of 7.
A typical pH probe would have specifications as shown in Table 1.
A pH probe plays an important role in this study, since data reliability will depend on the sensor’s accuracy and its stability. Two keys factors to consider when choosing a pH probe are its stability time after a temperature change and its stability time after a change in the buffer solution pH value. This is discussed in Jenway’s application note, “An Evaluation of Jenway Performance pH Electrode.”1
Aliquots of the pH 7 at 20°C and pH 4 at 60°C buffers were prepared. Each electrode was allowed to stabilize in the pH 7 buffer stirred at 200 rpm. The electrode was then rinsed with deionized water and transferred to the aliquot of the pH 4 buffer for a period of four minutes. The electrode was again rinsed with deionized water and returned to the pH 7 buffer. The time taken for the reading to remain stable for 10 seconds was then assessed. The test was repeated in triplicate for each probe. Tables 2 and 3 reveal the stabilization times.
Jenway’s performance compared to a general-purpose pH probe shows a difference up to a 50% faster response within the given conditions shown above. Using an instrument like this will benefit greatly from reducing the time required in analyzing the data because of its high throughput of samples.
Sensor Analog Signal-Conditioning Circuit
It’s important to understand the equivalent electrical diagram of the sensor probe to arrive at an appropriate signal-conditioning circuit. As described in the previous section, the pH probe consists of glass that creates an extremely high resistance that can range from 1 MΩ to 1 GΩ and acts as resistance in series with the pH voltage source (see figure).
Shown is a pH probe equivalent electrical circuit configuration.
Even a very small circuit current traveling through the high resistances of each component in the circuit (especially the measurement electrode’s glass membrane) will produce relatively substantial voltage drops across those resistances, seriously reducing the voltage seen by the meter. Making matters worse is the fact that the voltage differential generated by the measurement electrode is very small, in the millivolt range (ideally 59.16 mV per pH unit at room temperature). The meter used for this task must be very sensitive and have an extremely high input resistance.
Analog-to-Digital Conversion
For this type of application, given the response time of the sensor, the sampling rate for data gathering will now be an issue. With a given sensor resolution of 0.001 V rms and an analog-to-digital converter (ADC) full-scale voltage range of 1 V, a high-resolution ADC will not be required to achieve an effective resolution of 9.96 bits. Noise-free resolution is defined in units of bits with the equation:
Noise-free resolution = log2 [full-scale input voltage range/sensor peak-to-peak voltage output noise]
The sampling rate of the ADC can be a crucial factor for low-power application since the ADC’s sampling rate is directly related to its power consumption. So, given the sensor’s response time, the typical ADC sampling rate is able to be set into its lowest throughput. A microcontroller with an integrated ADC can be used to reduce component count.
Transceiver
Transmitting pH and temperature data requires a transceiver, and controlling a transceiver requires a microcontroller. Choosing both the transceiver and microcontroller involves some key considerations. When choosing a transceiver, they include:
- Operating frequency
- Maximum distance range
- Data rate
- Power consumption
- Licensing
Operating Frequency
In designing RF transmission, the operating frequency (OF) must determine if a sub-GHz or 2.4-GHz frequency will serve the application requirement. In an application that requires a high data rate and uses wide bandwidth like Bluetooth, the 2.4-GHz frequency is the best choice. But when the application is industrial, sub-GHz will be used because of the available proprietary protocols that readily provide the link layer of the network. Sub-GHz industrial, scientific, and medical (ISM) frequencies that proprietary systems primarily use are 433 MHz, 868 MHz, and 915 MHz.
Maximum Distance Range
Sub-1-GHz frequencies offer long-range capability that can accommodate high power and reach over 25 km. These frequencies, when used in a simple point-to-point or star topology, can effectively pass through walls and other barriers.
Data Rate
The data rate also needs to be determined as it affects the transmission distance capability and power consumption of the transceiver. A higher data rate consumes less power and can be used over a short distance while a lower data rate consumes more power and can be used in long-distance transmission. Increasing the data rate is a good method for improving power consumption, as it only draws bursts of current from the battery in short periods of time. However, this also reduces the distance of radio coverage.
Power Consumption
Transceiver power consumption is important for battery-powered applications. This is also a factor in many wireless applications because it dictates the data rate and distance range. The transceiver has two power-amplifier (PA) options to allow for greater usage flexibility. The single-ended PA can deliver up to +13 dBm of RF power, while the differential PA can provide up to +10 dBm. For illustration, Table 4 shows a summary of some of the PA output powers versus transceiver IDD current consumption. For completeness, the receive-mode current consumption is also shown.
Licensing
Sub-GHz includes license-free ISM bands at 433 MHz, 868 MHz, and 915 MHz. It’s widely used in industrial and highly suitable in a variety of wireless applications. It can be used in different regions of the world because this complies with the European ETSI EN300-220 regulation, the North American FCC Part 15 regulation, and other similar regulatory standards.
Erbe D. Reyta is a hardware applications engineer and Mark O. Ochoco is an application development engineer at Analog Devices Inc.
Reference
- Jenway Application Note, “An Evaluation of a Jenway Performance pH Electrode.”
About the Author
Erbe Reyta
Hardware Applications Engineer
Erbe D. Reyta has been a hardware applications engineer at Analog Devices Circuits from the Lab program for almost five years, where he has mainly focused on precision system hardware development. He earned his Bachelor’s in electronics and communications engineering at the University of the Philippines-Diliman. He is currently working toward his Master’s in computer engineering at Pamantasan ng Lungsod ng Maynila (University of the City of Manila) in the Philippines.
Mark Ochoco
www.analog.com
Mark O. Ochoco has been an application development engineer at Analog Devices for almost two years. His work mainly focuses on hardware and software development in the IoT field. Prior to joining ADI, he worked at Zilog for four years as a technical support and applications engineer focusing on application-specific embedded system on chip (SoC), both hardware and software. He earned his Bachelor’s in electronics and communications engineering at the Technological University of the Philippines-Manila.