2023 Nobel Prizes for Physics and Chemistry Have Direct EE Relevance
This article appeared in Electronic Design and has been published here with permission.
What you'll learn:
- For the Nobel in Physics, creation of attosecond laser pulses.
- For the Nobel in Chemistry, the discovery and synthesis of quantum dots.
The Nobel Prize awards from the Royal Swedish Academy of Sciences given in the “hard sciences” are sometimes awarded for abstract, hard-to-fathom advances, which may be fascinating but have little apparent immediate value. For example, the confirmation of gravity waves as predicted by Einstein led to a Physics prize in 2017, but the actual benefits of the impressive efforts are, thus far, only apparent for theory and research.
Other times, it takes decades for the benefits to appear. When Isidor Isaac Rabi discovered in the 1930s that protons could be twisted out of their normal orbital alignment via a magnetic field, and would then radiate RF when the field was removed (Nobel Prize, Physics 1944), the consensus was “interesting, but so what?”
It wasn’t until the 1970s, when Dr. Raymond Damadian realized and showed experimentally that there might be a way to use this atomic-level response to see the difference and create images. That’s because tumors contain more water and thus more hydrogen atoms than healthy tissue—and proved it by building the first crude MRI system.
In contrast, this year’s Nobel Prizes in physics and chemistry are much more practical and even have a relationship to mass production. Let’s look at these in more detail:
The Physics Prize
Pierre Agostini, Ferenc Krausz, and Anne L’Huillier received the award “for experimental methods that generate attosecond pulses of light for the study of electron dynamics in matter.” In brief, they devised a way to create attosecond laser pulses (one billionth of a billionth, or 10-18 second long). This is three orders of magnitude shorter (faster) than the femtosecond-long strobed laser pulses created in the 1980s by Ahmed Zewail at the California Institute of Technology (1999 Nobel Prize in Chemistry).
What are the potential uses for these attosecond pulses? They allow researchers to better capture the actions of an electron in motion, via a sort of “stop-action” imaging—and EEs are obviously interested in the secrets and flow of the electron. For example, if you direct attosecond laser pulses at a semiconductor, the material “snaps” almost instantaneously between blocking the flow of electrons to conducting them, potentially allowing for the production of ultra-fast electronic devices.
The technique could probe a variety of electron activities, including how the particles carry and block electric charge, how electrons bounce off one another, and how electrons collectively behave. Furthermore, Krausz—one of this year’s laureates—is also attempting to harness the power of attosecond pulses to detect subtle changes in blood cells that could indicate the early stages of cancer.
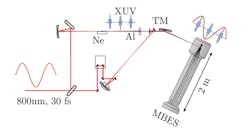
Generating Attosecond Pulses
So how does one create these attosecond pulses? If you shine a light on certain gases, their atoms will become excited and re-emit light overtones, which are additional colors of light at higher frequencies (shorter wavelengths) than the original. They found that for certain gases, such as argon, some of these overtones appeared brighter than others, but in an unexpected pattern.
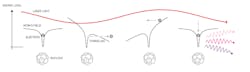
Using advanced quantum-mechanic principles, L’Huillier and other researchers were able to calculate the different intensities of the various overtones and so predict exactly how those atoms would, in turn, emit beams of quickly oscillating “extreme ultraviolet” light when a slowly oscillating infrared laser hit a cloud of atoms (high-harmonic generation, or HHG). They devised ways to overlay them so that these overtones added up to a new wave—one with peaks surging on the attosecond scale (Fig. 1).
Over the following years, Agostini and his group developed a technique called Rabbit, or “reconstruction of attosecond beating by interference of two-photon transitions.” With Rabbit, in 2001 Agostini’s group generated a string of laser pulses, each lasting 250 attoseconds (Fig. 2).
Concurrently, Krausz’s group used a slightly different method called “streaking” to produce and study individual bursts, each lasting 650 attoseconds. In 2003, L’Huillier and her colleagues created a laser pulse lasting just 170 attoseconds.
The Chemistry Prize
Moungi G. Bawendi, Louis E. Brus, and Alexei I. Ekimov were awarded the Nobel Prize in Chemistry for the discovery and synthesis of quantum dots. These nanoparticles are so tiny that their size determines their properties. These smallest components of nanotechnology now spread their light from televisions and LED lamps, and they can also guide surgeons when removing tumor tissue, among many other applications.
These precise nanocrystals consist of silicon and other semiconductor materials that are just a few nanometers wide—small enough to exhibit quantum properties just as individual atoms do, although they’re a hundred to a few thousand atoms in size. Because electrons can be trapped at certain energy levels within them, the nanocrystals can emit only certain wavelengths of light. By controlling the size of the particles, researchers can program precisely what color the quantum dots will flash when stimulated.
The wavelength of light they emit depends solely on the size of the particles. Electrons in larger particles have less energy and emit red light, whereas electrons in smaller particles have more energy and emit blue light. The specific color of light will emerge from the quantum dots and can be precisely controlled “simply” by regulating their size. That offers a huge advantage over the use of other kinds of fluorescent molecules, for which a new type of molecule is needed for every distinct color.
Quantum Dots Timeline
By the 1970s, physicists knew that quantum phenomena should in theory be associated with particles of extremely small size, just as they were with ultra-thin films, but that prediction seemed impossible to test: There seemed to be no good way of making and handling particles except inside other materials – and it would mask their properties.
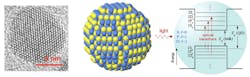
In 1981, while adding compounds of copper and chlorine to a glass, Eskimov found that the color of the glass depended entirely on the size of those added particles. He recognized that quantum effects were the likely explanation (Fig. 3).
In 1983 while at Bell Labs, Brus was running experiments on the use of light to drive chemical reactions. Brus (now at Columbia University) noticed that the size of nanoparticles also affected their optical properties even when they were floating freely in a liquid solution. Still, researchers were unable to precisely control the size and quality of these particles.
In 1993, however, Bawendi invented an “ingenious chemical method” for making perfect nanoparticles. He was able to control the exact moment in time when the crystals formed, and he was then able to stop and restart further growth in a controlled manner. His discovery made quantum dots widely useful in a variety of applications.
The above descriptions are obviously extreme simplifications of the scientific basis, technical accomplishments, collaborations, and historical timelines. Fortunately, the Royal Swedish Academy of Sciences has provided excellent additional support collateral, with both a more-general “popular science” paper and a highly technical “scientific background” paper for each category (see References below).
References
For Physics:
- Press release: The Nobel Prize in Physics 2023
- Popular science background: Electrons in pulses of light (pdf)
- Scientific Background: “For experimental methods that generate attosecond pulses of light for the study of electron dynamics in matter” (pdf)
For Chemistry:
About the Author

Bill Schweber
Contributing Editor
Bill Schweber is an electronics engineer who has written three textbooks on electronic communications systems, as well as hundreds of technical articles, opinion columns, and product features. In past roles, he worked as a technical website manager for multiple topic-specific sites for EE Times, as well as both the Executive Editor and Analog Editor at EDN.
At Analog Devices Inc., Bill was in marketing communications (public relations). As a result, he has been on both sides of the technical PR function, presenting company products, stories, and messages to the media and also as the recipient of these.
Prior to the MarCom role at Analog, Bill was associate editor of their respected technical journal and worked in their product marketing and applications engineering groups. Before those roles, he was at Instron Corp., doing hands-on analog- and power-circuit design and systems integration for materials-testing machine controls.
Bill has an MSEE (Univ. of Mass) and BSEE (Columbia Univ.), is a Registered Professional Engineer, and holds an Advanced Class amateur radio license. He has also planned, written, and presented online courses on a variety of engineering topics, including MOSFET basics, ADC selection, and driving LEDs.