October, 1967
Basic considerations and specific design steps are given for microwave-system refrigeration
The environmental effects on microwave hardware, when operated at cryogenic temperatures (4° K-77° K), must be taken into account in the design of circuit components and systems. The dimensions of a microwave component to be cooled must be such that its critical tolerances will be maintained under refrigeration.
While submersing a circuit in a cryogenic fluid may not appear difficult, especially for a “one-shot” experiment, problems arise when attempting to achieve a permanent, dependable, cooled microwave system. Principal problems to overcome in cryogenic cooling are moisture condensation and consequent freezing on waveguide walls (degrading rf transmission), changes in material dimensions with temperature (Fig. 1) and variations in thermal conductivity (Fig. 2).
The effects of moisture in a cooled microwave system can be virtually eliminated by removing moisture-bearing gas from the waveguide. This may be done in two ways: 1. By employing a “sealed” system where a moisture-free heat-exchange gas[1] or partial vacuum is present, or 2. By causing a moisture-free coolant gas to flow through the waveguide. The latter method is inherently less reliable because of its susceptibility to moisture condensation if and when the coolant supply is exhausted. This, along with the limited application of such a method, almost precludes its use in permanent cryogenic-temperature microwave systems.
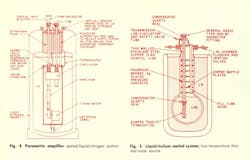
A “sealed” cryogenic microwave system requires hermetic integrity of the microwave circuitry throughout the thermal cycling. Also, the rf transmission properties of the waveguide and the desire for economical use of coolant may be significant concerns in designing the system. The system components are then designed accordingly.
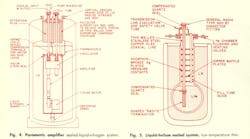
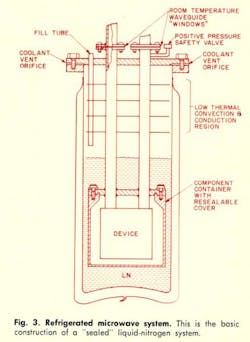
The basic major components of a “sealed” system (Fig. 3) would include: 1. The dewar flask, 2. Room temperature-waveguide windows, 3. A positive pressure safety valve, 4. A low-thermal-conductivity convection region, and 5. A resealable component or system container. It becomes apparent that the design and construction of cryogenic microwave system components involve several compromises between materials and methods. Design information for the basic components of a cooled system is presented in capsule form in the accompanying Cryogenic Design Table. Photographic views and line drawings of actual “sealed” systems, built for operation at liquid nitrogen (77° K) and liquid helium (4.2° K) temperatures are presented in Figs. 4, 5 and 6.
Coolant level indicator
While the cryogenic system is in operation, it may be necessary to monitor or sample the temperature at various points. While there are extensive low-temperature thermometry techniques, a simple carbon thermometer employing a stock carbon resistor may be used as a coolant level indicator. The resistance of carbon increases as its temperature decreases to a value which can be found experimentally. The resistor is located at the desired level and monitored during filling or operation.[2]
Safe system operation
In addition to familiarization with the correct handling procedures of cryogenic fluids, safe system operation in a cooled sealed system requires some form of positive-pressure safety valve. The safety valve is necessary to avoid the hazard created by a rapidly expanding volume of gas in a closed container. A leak in the system above the coolant level would allow moisture-laden air to be drawn in and possibly freeze the leak closed. As the defective system is then withdrawn from its dewar, the entrapped gas expands rapidly and the excess pressure within in turn causes the gas to expand even more violently. If the leak occurs below the coolant level, the gas generated by the cryogenic fluid trapped upon withdrawal will increase the internal pressure. The accompanying gas-to-liquid volume-ratio table gives the approximate gas-to-liquid volume ratio to be expected at standard temperature and pressure of liquid nitrogen, liquid air, and liquid helium.
In practice, a rubber stopper or plug (Fig. 7) provides a basic safety valve. Prior to evacuating the system, the plug is placed in position over the opening provided. A somewhat more complicated scheme must be employed if a heat-exchange gas is to be used within the system. In either case, the system must be completely checked for leaks prior to cooling to ensure system integrity and safety to personnel.
[1] The heat exchange gas must remain gas at the anticipated cooled temperature of operation. For example, dry helium gas would be used in a nitrogen-cooled system. However, nitrogen gas might liquefy in a helium-cooled system.
[2] For example, a 1-kΩ, ¼-W carbon resistance increases to ≈ 32 kΩ when immersed in liquid helium.