This file type includes high resolution graphics and schematics when applicable.
Editor's Note: Be sure to check out our interview with Glen Griffith, principal RF communications engineer for the Alfred Mann Foundation.
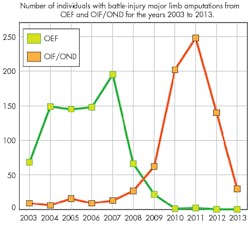
At the end of 2013, the total number of U.S. soldiers who suffered crippling battle-related amputations in the wars overseas exceeded 1500 (Fig. 1).1 Many of these soldiers suffered multiple limb amputations. Given the limitations of most current prosthetics, they were looking to a future that was bereft of most of the activities that formerly defined their lives. Particularly heartbreaking to many of these soldiers was the inability to return to a career that many felt was a life calling.
To both preserve the assets of injured career soldiers and improve their quality of life, organizations began investing in prosthetic technology development. From the Alfred E. Mann Foundation (AEMF) to DARPA, NIH, TATRC, the Walter Reed National Medical Center, Advanced Arm Dynamics, and Ottobock, these organizations strived to help a soldier live a healthy and active life post-injury—and potentially even continue to serve. As part of this effort, RF/microwave technology is being analyzed, reviewed, and even adopted to aid in next-generation prosthetics, which will soon function in ways indistinguishable from natural limbs.
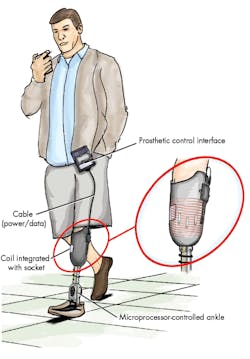
Before the invention and availability of the microprocessor-controlled prosthetic, there were very limited options for prosthetics that could be used throughout an active daily routine. This led to mostly application-specific prosthetics—for example, for running, walking, water use, or sports. Yet the microprocessor-controlled prosthesis allowed an adjustable interface to control the mechanical movers within the limb (Fig. 2).
Such enhanced limb rotation and control, which can be customized for the individual patient, enables much more lifelike and natural motion. By including joint programmability, developers added an extra layer of programming and patient training. Additionally, a power source and a controller for switching between different use modes were required. As a result, the patient now had to charge the prosthetic and adjust the programming of its microprocessor on a regular basis.
To ease this process, companies like Ottobock embraced wireless technology while adding usability and convenience features that would be prohibitive to implement otherwise. Their two main innovations were the wireless charging of the prosthetic battery and the wireless programming and mode switching of the microprocessor-controlled prosthesis rule-settings. Both of these clever uses of wireless technology remove the need for physical connectors in a prosthesis.
External connectors are common points of failure and environmental ingress. For its part, Ottobock developed the C-Leg, which employs inductive charging of the lower-limb prosthesis with a magnetic attachment at a point behind the knee. This attachment point functions as a wireless power receiver (Fig. 3). The inductive resonant coil behind the knee couples with a specialized charger, which is equipped with another resonant inductive coil. Charging lights and other indicators help to update the patient on the prosthesis’ battery condition.
Electronics within the C-Leg convert the low-megahertz wireless energy into direct current (DC) while a charging controller recharges the internal battery.
Because the charging system used with the C-Leg is inductive and resonant in nature, the prosthesis’ charging adapter can transfer power through clothing and even cosmesis. These cosmetic additions improve the aesthetics of the prosthesis. When converting the interconnect technology of a microprocessor-controlled prosthesis to wireless, the next step is to enable wireless programming and patient control of the various modes.
Originally, Ottobock used a USB Bluetooth transmitter connected to a PC. It communicated with a Bluetooth receiver wired to the prosthesis, also through a USB port. According to Philipp Kampas, program manager of lower extremity prosthetics for Ottobock, “The idea originated when patients ended up entangled in the cables during the setup process of the prosthesis. During initial configuration of the microprocessor knee, the patient had to walk around while connected by many wires to the prosthetist’s PC.
“We first used Bluetooth as an add-on device that replaced a serial communication cable for programming the microprocessor knee,” he continues. “As soon as this was established, it was obvious that an embedded Bluetooth receiver could also be used for the interface of the remote control.”
This solution solved many of the challenges stemming from the programming and mode setting of the microprocessor-controlled prosthesis. But the Bluetooth receiver also added an external component, which had its own set of failure modes and power requirements. The next stage of advancement was to incorporate wireless connectivity into the leg design. This revision of the wireless prosthesis omitted wired connections altogether. The redesign enabled a waterproof prosthetic leg that is essentially impervious to environmental ingress during regular activity.
The Genium and X3 lower-limb prosthesis are the result of this development. They use a Bluetooth communications link between a PC and the prosthesis. “We sell our products globally, and our patients travel around the world,” states Robert Hoffmann, head of Electronic Research & Development/Lower Extremities for Ottobock. “We needed a technology that could be used globally. Eleven years ago, there were not many options. This led to the use of the 2.45-GHz ISM band. The standard approach at the time was to use a proprietary amplitude-shift-keying (ASK) or frequency-shift-keying (FSK) single-frequency solution.
The key advantages of Bluetooth are the robust frequency-hopping scheme, the low-power consumption, and the support of the PC world.” Hoffmann adds, “The Bluetooth and inductive-charging technology are integral parts of the waterproof feature, since there are no more user-accessible connectors that are a potential weak spot for corrosion.”
Even with the complications and failure modes associated with the physical connections removed, integrating wireless technologies in a rugged and mobile platform made from moving conductive parts posed many challenges. Typically, prosthesis materials consist of aluminum, titanium, steel, or carbon-fiber composites, which are also conductive. Because an external antenna would reduce the rugged capabilities of the prosthesis, sophisticated designs and high-quality components were required.
To maintain high security with the wireless prosthesis’ connection, a custom-designed USB Bluetooth module for a PC is used to program the prosthesis. Specialized software is utilized to add another level of safety features, thereby ensuring correctly transmitted parameters.
A large number of soldiers have left the battlefield with amputations, leaving a palpable demand for prosthetics to become more lifelike and to integrate with technology that is familiar to the wearers. Hoffmann comments, “The next challenges are interfacing with mobile devices like Android and iOS smartphones. Also, reducing power consumption and optimizing RF and mechanical integration to get maximum transmission range are still issues. Another big problem is to find long-term available modules, because the product life cycles of medical devices are much longer than those of consumer products.”
Although wireless charging and programmability have enhanced many features of prosthesis technology, using sophisticated sensors with wireless capability promises to bring space-aged—and much hoped for—control of robotic limbs as prosthesis (Fig. 4).
This file type includes high resolution graphics and schematics when applicable.
Wireless Sensors Tap Muscle Nerves
This file type includes high resolution graphics and schematics when applicable.
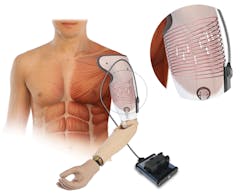
Together with with DARPA and other research-to-market organizations, the Alfred Mann Foundation (AEMF)—a transitional research engine that aids in bringing medical-device technology from research labs to commercial markets—is developing technology to enable sensors that will be implanted within muscle tissue. Such sensors will be able to wirelessly communicate with a robotic prosthesis worn by an amputee (Fig. 5). The implantable Myoelectric Sensor System (IMES) uses sensors embedded in residual muscle tissue in an amputated limb.
These sensors rely on the same control nerves as a natural limb for directing the analogous prosthesis. What enables this technology to work is the remote powering and communication performed by an in-sensor wireless module and a power-transmitting/receiving antenna within the prosthesis socket. Using embedded sensors improves the signal integrity and isolation significantly over current technology. Because other approaches use electromyographical (EMG) patches, which rest on the surface of the skin, they result in imprecise nerve signals.
The myoelectric sensor module is composed of a ceramic cylinder with biocompatible metallic end-caps. The end-caps contain the custom electronics for both the EMG signal sensing and module also houses the RF functions (Fig. 6). A custom application-specific integrated circuit (ASIC) within the sensor module is used to convert the analog EMG signals into a transmissible sampling. The present technology can transmit a 121-kHz data link that can adequately control a robotic arm for the initial subject.2 The initial telemetry design used a 6.78-MHz signal that, in theory, could transmit a full-bandwidth EMG signal. As a product of phase-coherence issues, errors beyond a usable level occurred. As a result, the researchers opted for a lower-bandwidth and sampled EMG signal.
Each IMES device amplifies, filters, rectifies, and integrates the EMG signal based on a calibration-initialization process. That process is performed after insertion and stable incorporation into the body. The RF module within each sensor is capable of bidirectional telemetry, as is the transceiver coil. That coil is designed to be laminated in place within a prosthetic socket frame.
The transmitted EMG data is received by the external coil, where it is digitized and transmitted to the external prosthetic controller interface (PCI). The system is capable of processing multiple EMG signal channels and retransmitting those to the PCI. The multiple EMG signals can be used to reconstruct different motor-control signals, which reproduce the dominant motion capability of a real limb.
The PCI is capable of splitting the multiple EMG digital data streams from the external coil and reconstructing the digitally-converted data back into analog data. The data then goes through another round of filtering. Next, the reconstructed and filtered analog signal is sent to the motor controller for the specific nerve tapped by the IMES. In this approach, multiple motor actions can occur simultaneously, which enables complex motor-control motions like pick-and-place and wrist pivoting while extending or contracting multiple fingers.
The first IMES system implemented by the group offered three simultaneous degrees of freedom using six separate sensors for a transradial amputation of a patient’s arm. The Touch Bionics i-limb ultra-hand enabled the patient to control wrist pronation/supination, hand open/close, and thumb abduct/adduct.
To extend the technologies usefulness, plans are in place to use the IMES technology with patients that have undergone targeted muscle reinnervation (TMR) surgery. TMR surgery remaps the nerves that are used for more external limb-control functions to less external limb nerve sites. Using an IMES on this remapped nerve site would enable amputees with little limb remaining to use a bionic prosthesis (Fig. 7). The AEMF is working with the Ottobock DynamicArm for TMR control in conjunction with the IMES. This solution is currently under regulatory review for a clinical feasibility study in the EU. Additionally, the foundation is working with Ossur to implement lower-limb prostheses for ankle movement.
Future enhancements include the addition of a sensor on a leaded IMES (LIMES), which can be used to target nerve-control centers beyond a feasible distance from a prosthesis socket. This method would enable an IMES to target nerve sites up to 12 inches away from the power transfer and telemetry zone of the external coil. The LIMES may also simplify the design of the external coil, allowing a standard coil design while preventing the need for custom coils that are socket-and-prosthetic dependent.
As the commercial-wireless and defense markets continue to invest in advancing wireless technology, more options will emerge to overcome control, sensing, and communications challenges in many industries. With the benefits of access to sensor data closer to the source of the information—and without degradation from physical cables—the market for more low-cost and small-form-factor wireless sensor nodes will increase.
Many expect the Internet of Things (IoT) to be used mainly for convenience technologies like smart homes, fitness tracking, and smartphone control of everyday things. But there are many more uses of the base technologies enabling the IoT, such as prosthetic limb control, remote machine control in biohazard/harsh environments, and exploring the world around us in ever-increasing detail.
Resources
This file type includes high resolution graphics and schematics when applicable.